Engineers
Experimental Plans
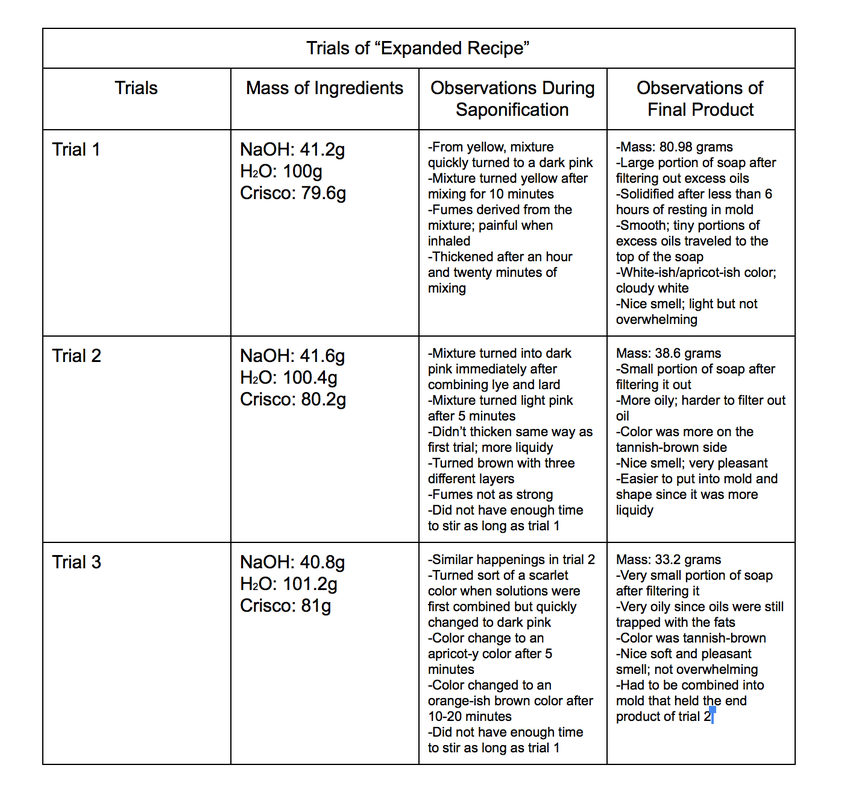
- Using a triple beam balance, measure the mass of a 140mL beaker. Find the theoretical sum of the beaker and 80g of Crisco.
- With a scapula or spoon, add Crisco into the beaker until the balance reads the theoretical sum of the two masses.
- Remove beaker with Crisco from the balance and place a 250 mL beaker onto the beaker. Find the theoretical sum of the beaker and 100g of water.
- Add water into the beaker until the balance reads the theoretical sum of the two masses.
- Remove beaker with water from balance and replace with another empty 140mL beaker. Measure mass of beaker.
- Using a calculator, find the theoretical sum of the masses of both the beaker and 40.8g of sodium hydroxide. Then with a scapula, carefully add sodium hydroxide into beaker until the balance reads the theoretical sum of both of the masses.
- Remove beaker with sodium hydroxide off from the balance.
- Slowly add the sodium hydroxide into the beaker with water while stirring the mix at the same time with a stirring rod. Stir until all crystals dissolve so that it turns into a lye mixture.
- Set beaker with Crisco onto a hot plate with its heat set at 5. Let the lard melt into oil. Stir when necessary. Remove from hot plate when the lard is completely melted.
- With the hot plate set at 3, place lye mixture on top. Slowly and carefully add melted lard into the lye mixture while stirring contents together at the same time.
- Continue to stir contents at a careful yet steady pace. Remove from heat source when contents start to boil for a minute or two then put back onto hot plate.
- When contents start to thicken, add 5 grams of scented oil into mixture. Continue to stir until the top layer has thickened, sort of like a paste.
- Filter mixture by placing and securing a paper towel over an empty 250mL beaker and pouring mixture onto paper towel to separate the molten soap and the oil. Repeat this twice to remove all excess oils.
- Set molten soap into a mold. Cover top to secure heat and let sit for 24-48 hours to solidify. Remove from container and use to wash hands after 24-48 hours.
Stoichiometric Calculations
Initial Recipe Percent Composition
2.5g Sodium Hydroxide 12.0%
3.62g Distilled Water 17.3%
14.8g Crisco (Lard) 70.7%
Total mass: 20.92g or ~21g
Lye Mixture: Percent Composition (According to Initial Recipe)
2.5g Sodium Hydroxide 12.0%
3.62g Distilled Water 17.3%
14.8g Crisco (Lard) 70.7%
Total mass: 20.92g or ~21g
Lye Mixture: Percent Composition (According to Initial Recipe)
- Water: 59.2%
- Sodium Hydroxide: 40.8%
Lab Apparatus Design
-NaOH (lye tablets) 54.32 g
-Water 79.08 g
-Lard 321.48 g
-Vanilla scents (Sandalwood Vanilla, Kitchen Spice scents) 5g each
-Beakers
-Balance
-Glass stir Rod
-Hot plate
-Coffee filters/paper towels
-Thermometer
-Mold-oval shaped
-Water 79.08 g
-Lard 321.48 g
-Vanilla scents (Sandalwood Vanilla, Kitchen Spice scents) 5g each
-Beakers
-Balance
-Glass stir Rod
-Hot plate
-Coffee filters/paper towels
-Thermometer
-Mold-oval shaped
Standards Operating Procedure/Controls
Cleaning
● All lab equipment must be washed thoroughly and appropriate as instructed.
● The last 7 minutes of all lab endeavors MUST always be reserved for lab cleanup.
Clothing
● Confine long hair during laboratory activities; it is a fire and contamination hazard.
● Secure dangling jewelry and loose or baggy clothing that could get caught on
equipment.
● Roll long sleeves above the wrist. .
● Lab aprons have been provided for your protection and should be worn during laboratory
work.
● Wear appropriate eye protection whenever you are working in the laboratory. Safety
goggles must be worn during hazardous activities involving caustic/corrosive chemicals,
heating of liquids, and other activities that may injure the eyes.
● Splashes and fumes from hazardous chemicals present a special danger to wearers of
contact lenses.
● Employees with open skin wounds on hands must wear gloves.
● Read summarized material safety info of chemicals in use.
General
● Conduct yourself in a responsible manner at all times in the laboratory.
● Never work alone; a supervisor must be present during the performance of all laboratory
work.
● Prepare for each laboratory activity by following through with research and preparation
before entering the lab. Follow all regulations implicitly.
● Make a note of all verbal or written modifications made by upper management.
● Know the proper fire and earthquake procedures. In an emergency or drill, containers
must be closed, gas valves turned off, fume hoods turned off, and any electrical
equipment turned off.
● Work areas should be kept clean and tidy at all times.
● To prevent injury from chemical spills, stand (do not sit) at lab stations when using
caustic or hazardous chemicals.
● Know the locations and operating procedures of all safety equipment including the first
aid kit, eyewash station, fire extinguisher, fire blanket, fire alarm, gas shut off, phone,
and exits.
● Eating or drinking, of any kind, is not permitted in the laboratory room at any time
● All lab equipment must be washed thoroughly and appropriate as instructed.
● The last 7 minutes of all lab endeavors MUST always be reserved for lab cleanup.
Clothing
● Confine long hair during laboratory activities; it is a fire and contamination hazard.
● Secure dangling jewelry and loose or baggy clothing that could get caught on
equipment.
● Roll long sleeves above the wrist. .
● Lab aprons have been provided for your protection and should be worn during laboratory
work.
● Wear appropriate eye protection whenever you are working in the laboratory. Safety
goggles must be worn during hazardous activities involving caustic/corrosive chemicals,
heating of liquids, and other activities that may injure the eyes.
● Splashes and fumes from hazardous chemicals present a special danger to wearers of
contact lenses.
● Employees with open skin wounds on hands must wear gloves.
● Read summarized material safety info of chemicals in use.
General
● Conduct yourself in a responsible manner at all times in the laboratory.
● Never work alone; a supervisor must be present during the performance of all laboratory
work.
● Prepare for each laboratory activity by following through with research and preparation
before entering the lab. Follow all regulations implicitly.
● Make a note of all verbal or written modifications made by upper management.
● Know the proper fire and earthquake procedures. In an emergency or drill, containers
must be closed, gas valves turned off, fume hoods turned off, and any electrical
equipment turned off.
● Work areas should be kept clean and tidy at all times.
● To prevent injury from chemical spills, stand (do not sit) at lab stations when using
caustic or hazardous chemicals.
● Know the locations and operating procedures of all safety equipment including the first
aid kit, eyewash station, fire extinguisher, fire blanket, fire alarm, gas shut off, phone,
and exits.
● Eating or drinking, of any kind, is not permitted in the laboratory room at any time
Data
Data Analysis
Conclusion
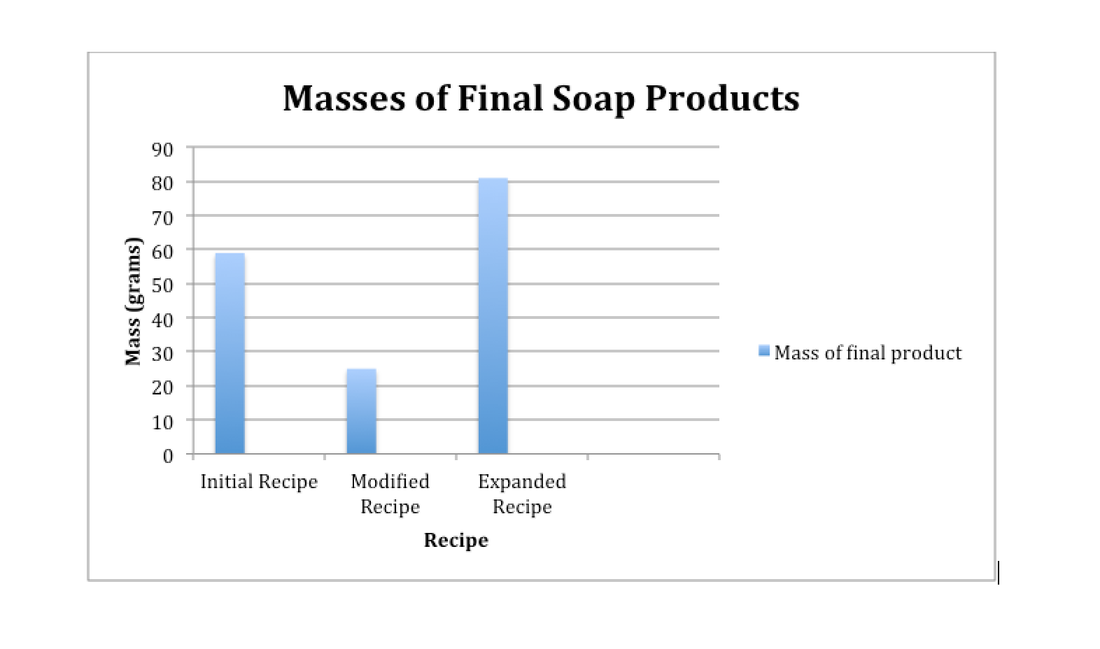
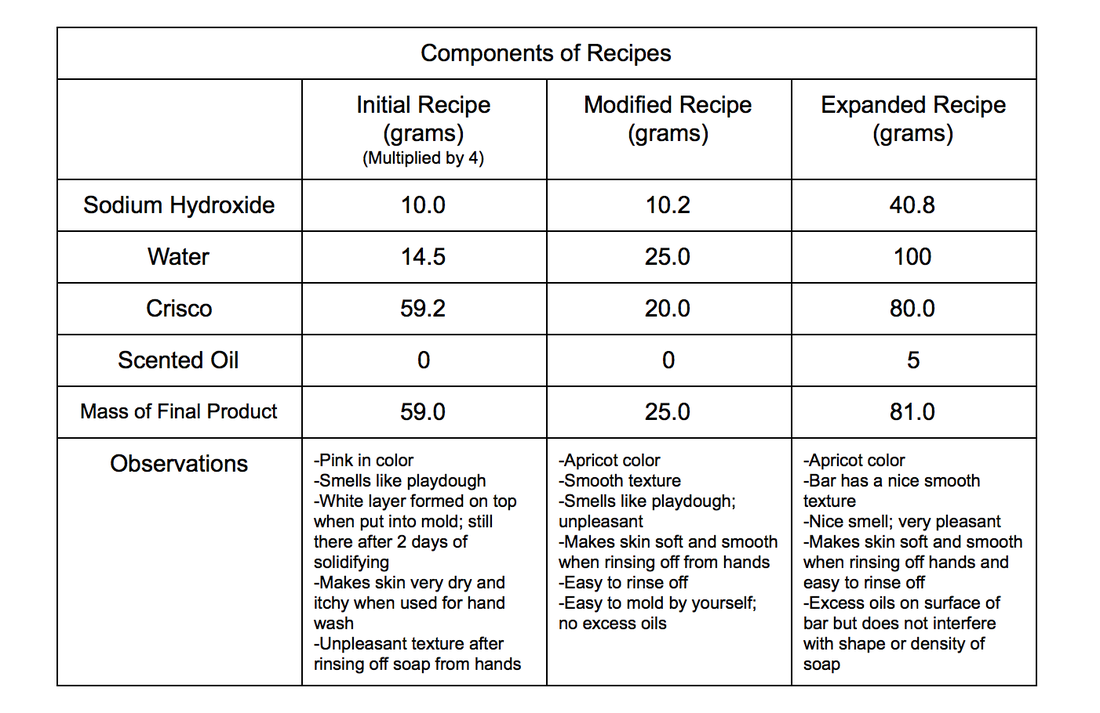
The different soap bars made gave the idea of the final recipe to make the perfect soap bar: The Expanded recipe.
With the expanded recipe, the soap created had good texture, consistency, and tender to hands. The soap made did its job of cleansing hands, as well as not drying them out. It also made the most product, producing 81.0 grams of soap. Although the trials were not consistent when making the Expanded recipe, the masses all came out to about the same, which means that the recipe can be used for mass reproduction.
These soap products created were made to match the demands of local Ewa residents. The scent, color, and effectiveness all matched the standards of the community.
With the expanded recipe, the soap created had good texture, consistency, and tender to hands. The soap made did its job of cleansing hands, as well as not drying them out. It also made the most product, producing 81.0 grams of soap. Although the trials were not consistent when making the Expanded recipe, the masses all came out to about the same, which means that the recipe can be used for mass reproduction.
These soap products created were made to match the demands of local Ewa residents. The scent, color, and effectiveness all matched the standards of the community.